Table of Contents
Heat Treatment Introduction
Heat treatment is a controlled operation that is used to modify the physical and mechanical properties of materials, primarily metals and alloys.
It alters the microstructure of the material to achieve desired characteristics like increased strength, hardness, ductility, and wear resistance and corrosion.
- It involves heating the material to a specific temperature and controlled cooling rates, often using different mediums like air, oil, or water.

Fig. Heat Treatment
- It modifies the internal structure of atoms within the material, including the size and arrangement of grains and phases.
- Heat treatment is widely used in various industries, including manufacturing, aerospace, and automotive, to enhance material properties and performance.
- According to J.F. Heat Treating Inc., the practice of heat treating metals has been around since ancient times, when primitive societies used crude methods to alter the properties of metals.
- Heat treatment is also a versatile tool for tailoring material properties to meet specific application requirements.
Definition
- Heat treatment is the process of heating and cooling a metal or alloy by using specific predetermined methods for the purpose of obtaining a certain desired shape and properties of the metal.
- The process of converting the solid metal into desired shape by heating is known as heat treatment process.
- The properties of a metal depend upon its structure.
Heat Treatment Process
- The heat treatment process is a series of operations involving the heating and cooling of metals in the solid state.
It involves heating the material or a sample to a suitable temperature, holding it there for a period, and then cooling it down at different rates.
This process modifies the material’s microstructure, which affects its properties like hardness, strength, ductility, and toughness.
Objectives of Heat Treatment
It increases the strength, hardness, and wear resistance of the materials.
It changes the structure and size of grain.
It increases the ductility and softness of the materials.
It improves machinability and corrosion resistance.
It relieved the internal stress.
It enhances altering electrical and magnetic properties.
- It increases the toughness of metals.
It improves the cutting properties of the materials.
Stages of Heat Treatment
- There are following Stages in heat treatment:
Stage 1
- Warming the metal gradually to guarantee a uniform temperature.
Stage 2
- Drenching ( Holding ) the metal at a given temperature for a given time frame.
- In Soaking, internal structure changes take place.
- Splashing period relies upon the compound investigation of the metal and the mass of the part.
Stage 3
- Cooling the metal to room temperature.
- Cool the metal in direct contact with a Cooling Medium composed of a gas, liquid, solid, or combination of these.
Heat Treatment of Steel
- Steel can be heat treated to produce a great variety of micro structures and properties hence, it is a most popular solid metal.
- It is an alloy of iron and carbon, which may contribute upto 2.1 % of its weight. Steels are a large family of metals.
- Every one of them are amalgams in which, iron is blended in with carbon and other component.
- Steel is fundamentally an alloy of iron and carbon, with the the carbon content varying up to 1.5 %.
- Steels are of different types as follows.
A. Dead mild steel
B. Mild steel
C. Medium carbon steel
D. High carbon steel
Also read:
Structure of steel
- The shape of steel depends upon the structure of iron or different types of molecules and quantity of carbon in it.
There are many types of steel structure are as follows.
1. Ferrite
- It is a chemical compound of ceramic materials with iron i.e., Fe 2 O 3.
- It is almost pure state of an iron due to the presence of 0% carbon in it.
- They can’t be hardened by rapid cooling.
- It is soft, ductile and comparatively weak.
- It has more magnetic property.

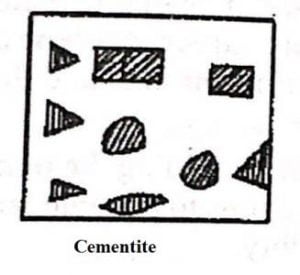
2. Cementite
- When carbon exists in steel as a chemical compound it is called iron carbide ( Fe 3 C ) Or Cementite.
- This alloy is very hard and brittle but it is not strong.
- It increases with more percentage of carbon.
- It shows magnetic property below 250 ° C.
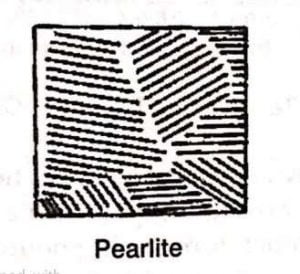
3. Eutectoid / Pearlite steel
- It is the mechanical aggregate of ferrite and cementite in equilibrium containing 0.88% carbon.
- It is made up of alternate white and dark bonds.
- The size of the pearlite depends upon rate of cooling.
- The rapid cooling will lead to the formation of the pear lite.
- The strength of the iron and steel is due to pearlite.
- It is much stronger than ferrite or cementite.
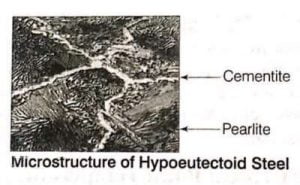
4. Hypoeutectoid Steel
- Hypoeutectoid steel consists of pearlite and cementite at room temperature.
- It contains carbon more than 0.8%.
- When slowly cooling a hypoeutectoid steel, the cementite will begin to crystallise first.
- When the steel remaining becomes eutectoid in composition, it will crystallise into pearlite. Since, cementite is much harder than pearlite.
Effect of Heat Treatment on Internal Structure of Steel
- To change the internal structure of steel, applying the heat treatment in a furnace.
- The internal changes depend upon the heating temperature conditions.
- If a specimen of the steel is heated, then its temperature increases continuously upto 723°C but after reaching the temperature 723°C, it remains same for some time.
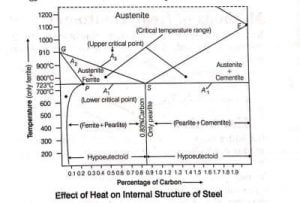
- Thereafter, it increases slowly and reached at certain temperature, then it again increases at the same rate as before.
- After reaching the temperature of 723°C , the heat energy consumption by specimen of steel becomes maximum which is further used in changing the internal structure of the steel and remaining part of the heat energy is used to increase the temperature of the steel.
- In this process, the lower critical point ( LCP ) of all carbon quantities has same temperature i.e., at 723°C but its Upper Critical Point ( UCP ) always vary.
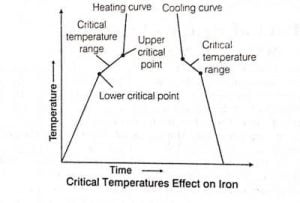
Critical Temperature
- The basic temperature of a substance is the temperature at or more which fume of the substance can’t be melted.
- There are mainly two states of the critical temperature, which are as follows:-
i. Lower critical point temperature
- The temperature at which the structure of steel to be an austenite ( i.e., at 723°C ), is called the lower critical temperature for all plain carbon steels.
- Normally, the value of lower critical temperature for all types of steel are 723°C but upper critical temperature is changeable.
ii. Upper critical point temperature
- The temperature at which the structure of steel completely changes to austenite is called the upper critical temperature.
- This varies depending on the percentages of carbon in steel.
Methods of Heat Treatment
- There are 9 methods of Heat treatment which are named as:-
1. Annealing ( Furnace cool )
2. Normalising ( Air cool )
3. Hardening or Quenching
4. Tempering
5. Case Hardening
6. Surface Hardening
7. Austempering
8. Marquenching
9. Ausforming
1. Annealing ( Furnace cool )
- Annealing is a heat treatment process which involves the heating of steel sample above critical temperature ( 727 degree ) and holding it there for about 1 hours so that their complete austenization take place.
- Then, it allow to cool slowly in the furnace.
- It is also known as Furnace cooling process.
Purposes of Annealing
i. To improve the ductility of the sample.
ii. It enhances the mach-inability of the sample.
iii. It reduces the internal resting force ( internal stress ).
iv. To reduce or eliminate structural in homogeneity.
2. Normalising
- Normalising is a process which is similar to annealing process. Here, the specimen is heating beyond the upper – critical limit temperature ( 227-912 ) degree while lower critical temperature ( 227 ) degree and cool in open air.
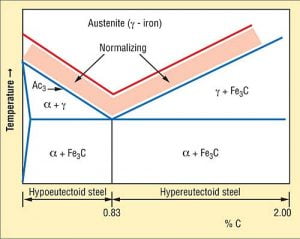
Graph.1 Normalising process
- In this process, the rate of cooling will be more as compare to annealing process.
- The strength of sample as well as its mach inability is enhance.
- It is applicable for ferrous metals only.
Purposes of Normalising
- To improve structure of weld.
- To soften metal.
- Refine grain structure.
- To prepare steel for sub heat treatment.
3. Hardening or Quenching

Image- Quenching
- In this procedure, steel is cooling in water and oil showers.
- The rate of cooling is very fast as compare to annealing and normalising process.
- The hardness of a metal is legitimately relative to the uni – pivotal yield worry at the area of the forced strain.
- It reduces the mach inability of the sample.
4. Tempering
- This process is using to increase the mach – inability of harden steel.
- In this procedure, test is warming underneath the lower basic temperature ( 227 ) degree and holding it for quite a while and afterward cooling it gradually.
Purpose of Tempering
- To soothe the steel from inner anxieties and strains.
- To regulate the hardness and toughness.
- Reduce the brittleness.
- To restore some ductility.
- To reduce shock resistance.
5. Case Hardening
- It is generally call as substance heat treatment process.
- In this process, steel surface is processing by the addition of carbon.
Types of Case Hardening
A. Carburizing
B. Nitriding
C. Cyaniding
D. Induction hardening
E. Flame hardening
A. Carburising
- In this process, steel is heating in contact with charcoal,wood.
- After heating, it will be followed by rapid cooling.
B. Nitriding
- Nitriding is a heat treatment process in which steel will heating in atmosphere of ammonia gas ( NH3 ) at the temperature ( 1|2 – 1|3 ) carburizing process.
- At this temperature NH3 gas dissociate in nascent nitrogen and hydrogen.
- After this nitrogen will get diffused into steel in type of nit – rides.
Advantages of Nitriding
- High fatigue life.
- High hardness than carburised and hardened components.
- Better corrosion resistance than carburised and hardened components.
Disadvantages of Nitriding
- No heat treatment can be done after nitriding.
- White layer.
- Thin case depth.
C. Cyaniding
- Cyaniding is a procedure wherein steel test is warmed in air of sodium cyanide.
- Both carbon and nitrogen will get diffused into steel test.
- The nitrides are structure during heat treatment and cyanides are structure during cooling.
D. Induction Hardening
- This process is applicable to high carbon steel.
- In this process, steel sample is heating to red hot condition by induction hardening.
- Then steel sample is quenching into water.
Advantages of Induction Hardening
- Increase fatigue strength.
- Localized areas can be heat treated.
- Low operating costs.
- May be incorporate into cell manufacture.
Disadvantages of Induction Hardening
- High capital investment.
- Only certain steels can be induction hardened.
E. Flame Hardening
- Fire solidifying is a surface solidifying technique that includes warming a metal with a high temperature fire, trailed by extinguishing.
- It is utilized on medium carbon, gentle or compound steels or cast iron to create a hard, wear – safe surface.
Benefits of Flame hardening
- Increase wear resistance.
- Reduce processing time.
- To reduce cost by hardening only selective areas.
- Less distortion.
- Ability to use low to medium carbon steels.
6. Surface Hardening
- Surface hardening is process in which treatment of steel by heat or mechanical means to increase the hardness of the outer surface while the core remains relatively soft.
- Its also valuable for its low and superior flexibility in manufacturing.
7. Austempering
- Austempering is applicable for ferrous metals, most notably steel and ductile iron.
- In steel it creates a bainite miniaturized scale structure where as in cast iron it delivers a structure of a roundabout ferrite and high carbon, balanced out austenite known as ausferrite.
- It is primarily use to improve mechanical properties or reduce / eliminate distortion.
Advantages of Austempering
- Bainite properties are same of martensite and pearlite and excessively like tempered Martensite.
- Structure and properties are transitional to tempered martenites without including in martensitic change.
- Dimensional security is progressively because of nonattendance of held austenite.
Disadvantages of Austempering
- Hardness is not high.
- Quite expensive.
- Applicable to high harden ability steels.
8. Marquenching
- It permits the transformation of Austenite to Martensite to take place at the same time through out the structure of the metal part.
- Martempering is otherwise called ventured extinguishing or interfered with extinguishing.
- During this process residual stresses are developed.
- Its also reduce susceptibility to cracking.
9. Ausforming
- It is also known as low and high temperature thermomechanical heat treatment.
- It is a technique used to expand the hardness and sturdiness of a compound by all the while hardening, fast cooling, twisting and extinguishing to change its shape and refine the microstructure.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Thanks for reading. If the articles help you then spread your love and don’t forget to share it.
Very good explanation sir..
Thanks so much sir
Thanks….
You explain very well sir 🤗🤗
Thanks…